药品详情:
【齐拉西酮Ziprasidone 简述】
齐拉西酮是一种非典型抗精神病药,其结构与吩噻嗪类或丁酞苯类抗精神病药物不同。体外研究显示,齐拉西酮对多巴胺D2, D3, 5-羟色胺5HT2A, 5HT2C、5HT1A、5HT1D、a-肾上腺素能受体具有较高的亲和力,对组胺H1受体具有中等亲和力,对包括M胆碱能受体在内的其他受试受体/结合位点未见亲和力。



【齐拉西酮Ziprasidone 适应症】
本品适用于治疗精神分裂症患者急性激越症状。根据最新临床经验,盐酸齐拉西酮胶囊对强迫性神经症同样有显著疗效。
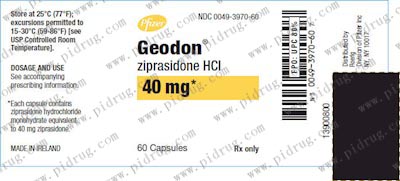
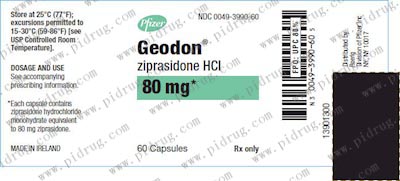
【齐拉西酮Ziprasidone 规格】
本品为胶囊装,每粒含有效成份20mg/40mg/60mg/80mg,每瓶60片装。
本品亦有注射剂,规格为20mg/mL。
【齐拉西酮Ziprasidone 服用方法】
推荐剂量为每日10-20mg,最大剂量为每日40mg。
【齐拉西酮Ziprasidone 注意事项】
国外研究表明:单剂肌注齐拉西酮的生物利用度为100%,达峰时间为60分钟或更早,平均半衰期(T1/2)为2-6小时。采用增加剂量方式和连续肌注3天观察,未出现蓄积。尽管对肌注齐拉西酮的代谢和消除未作系统评价,肌注齐拉西酮应与其口服制剂的代谢途径相同。
Ziprasidone
Ziprasidone, sold under the brand name Geodon among others, is an atypical antipsychotic that is used for the treatment of schizophrenia as well as acute mania and mixed states associated with bipolar disorder.[3] Its immediate release intramuscular injection form is approved for acute agitation in people with schizophrenia. Ziprasidone is also used off-label for depression, bipolar maintenance, and post-traumatic stress disorder (PTSD).[4]
Common side effects include dizziness, drowsiness, dry mouth, and twitches.[5][6]Although it can also cause weight gain, the risk is much lower than for other antipsychotics.[7]
The by mouth form of ziprasidone is the hydrochloride salt, ziprasidone hydrochloride. The intramuscular form is the mesylate rather than hydrochloride salt, ziprasidone mesylate trihydrate, and is provided as a lyophilized powder. Ziprasidone gained approval in the United States on February 5, 2001.[8][9]
Uses
Ziprasidone is approved by the U.S. Food and Drug Administration (FDA) for the treatment of schizophrenia as well as acute mania and mixed states associated with bipolar disorder. Its intramuscular injection form is approved for acute agitation in schizophrenic patients for whom treatment with just ziprasidone is appropriate.[10]
In a 2013 study in a comparison of 15 antipsychotic drugs in effectiveness in treating schizophrenic symptoms, ziprasidone demonstrated mild-standard effectiveness. 15% more effective than lurasidone and iloperidone, approximately as effective as chlorpromazine and asenapine, and 9-13% less effective than haloperidol, quetiapine, and aripiprazole.[11] Ziprasidone is effective in the treatment of schizophrenia, though evidence from the CATIE trials suggests it is less effective than olanzapine, and equally as effective compared to quetiapine. There are higher discontinuation rates for lower doses of ziprasidone, which are also less effective than higher doses.[12]
Adverse effects
Ziprasidone (and all other second generation antipsychotics (SGAs)) received a black box warning due to increased mortality in elderly patients with dementia-related psychosis.[13]
Sleepiness and headache are very common adverse effects (>10%).[5][6]
Common adverse effects (1–10%), include producing too much saliva or having dry mouth, runny nose, respiratory disorders or coughing, nausea and vomiting, stomach aches, constipation or diarrhea, loss of appetite, weight gain (but the smallest risk for weight gain compared to other antipsychotics[7]), rashes, fast heart beats, blood pressure falling when standing up quickly, muscle pain, weakness, twitches, dizziness, and anxiety.[5][6] Extrapyramidal symptoms are also common and include tremor, dystonia (sustained or repetitive muscle contractions), akathisia (the feeling of a need to be in motion), parkinsonism, and muscle rigidity; in a 2013 meta-analysis of 15 antipsychotic drugs, ziprasidone ranked 8th for such side effects.[14]
Ziprasidone is known to cause activation into mania in some bipolar patients.[15][16][17]
This medication can cause birth defects, according to animal studies, although this side effect has not been confirmed in humans.[13]
Recently, the FDA required the manufacturers of some atypical antipsychotics to include a warning about the risk of hyperglycemia and Type II diabetes with atypical antipsychotics. Some evidence suggests that ziprasidone does not cause insulin resistance to the degree of other atypical antipsychotics, such as Zyprexa. Weight gain is also less of a concern with ziprasidone compared to other atypical antipsychotics.[18][19][20][21] In fact, in a trial of long term therapy with ziprasidone, overweight patients (BMI > 27) actually had a mean weight loss overall.[13]According to the manufacturer insert, ziprasidone caused an average weight gain of 2.2 kg (4.8 lbs), which is significantly lower than other atypical antipsychotics, making this medication better for patients that are concerned about their weight. In December 2014, the FDA warned that ziprasidone could cause a potentially fatal skin reaction, Drug Reaction with Eosinophilia and Systemic Symptoms, although this was believed to occur only rarely.[22]
注:药品如有新包装,以新包装为准。以上资讯来源于网络或由高等医药院校的学生志愿者翻译(如有错漏,请帮忙指正),仅供医护人员内部讨论,不作任何用药依据,具体用药指引,请咨询主治医师。
如您发现本网站有文字编辑或内容错误,请点击此处发送(需要安装有foxmail或outlook支持),
或发邮件至:info@pidrug.com,香港济民药业感谢您的到访!
欢迎您添加香港济民药业微信,或在公众号内留言。







